copper chromate
copper chromate
CAS: 13548-42-0
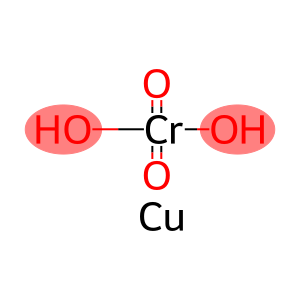
Molecular Formula: CrH2O4.Cu
copper chromate - Names and Identifiers
| Name | copper chromate |
| Synonyms | copper chromate COPPER(II)CHROMATE(VI) |
| CAS | 13548-42-0 |
| EINECS | 236-922-7 |
copper chromate - Physico-chemical Properties
| Molecular Formula | CrH2O4.Cu |
| Molar Mass | 181.56 |
| Melting Point | decomposes at 400℃ [KIR78] |
| Water Solubility | mol/L solution H2O: 0.0020 (25°C); solid phase, CuCrO4 ·H2O [KRU93] |
| Appearance | red-brown crystals |
copper chromate - References
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| production method | preparation method a solution is prepared by dissolving 4.5g of sodium chromate in 20ml of water, this solution was added with cooling to a solution of 17g of copper carbonate CuCO 3 in 30ml of water containing 30g of chromium trioxide CrO 3. The resulting red-brown precipitate was filtered off and washed with cold water. The resulting dihydrate was placed under reduced pressure even in a desiccator. The crystal water is also not easily lost. |
| category | toxic substances |
| explosive hazard characteristics | spontaneous combustion by reaction with sulfide |
| flammability hazard characteristics | non-combustible; Toxic chromium-containing, copper compound smoke generated from fire scene |
| storage and transportation characteristics | warehouse low temperature, ventilation, dry; Fire prevention; High temperature protection |
| fire extinguishing agent | water, carbon dioxide, dry powder, foam |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: copper chromate Request for quotation
CAS: 13548-42-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13548-42-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: copper chromate Request for quotation
CAS: 13548-42-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 13548-42-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
copper chromate
4-氯代硫代苯甲酰胺
Cyclohexanone, 5-methyl-2-(1-methyl-1-phenylethyl)-
叔-丁基 3-甲酰基-3-甲基吖丁啶-1-甲酸基酯
Methyl 2,5-dicyanobenzoate
1,2,3,4-四氢-6-三氟甲基异喹啉
34063-53-1
Trans-4-Propylcyclohexyl-Benzene
158930-09-7
4-(三氟甲基)哌啶盐酸盐
4-氯代硫代苯甲酰胺
Cyclohexanone, 5-methyl-2-(1-methyl-1-phenylethyl)-
叔-丁基 3-甲酰基-3-甲基吖丁啶-1-甲酸基酯
Methyl 2,5-dicyanobenzoate
1,2,3,4-四氢-6-三氟甲基异喹啉
34063-53-1
Trans-4-Propylcyclohexyl-Benzene
158930-09-7
4-(三氟甲基)哌啶盐酸盐
